A print-and-fuse Strategy for Sacrificial Filaments Enables Biomimetically Structured Perfusable Microvascular Networks with Functional Endothelium Inside 3D Hydrogels
Matthias Ryma, Hatice Genç, Ali Nadernezhad, Ilona Paulus, Dominik Schneidereit, Oliver Friedrich, Kristina Andelovic, Stefan Lyer, Christoph Alexiou, Iwona Cicha, Jürgen Groll
Abstract
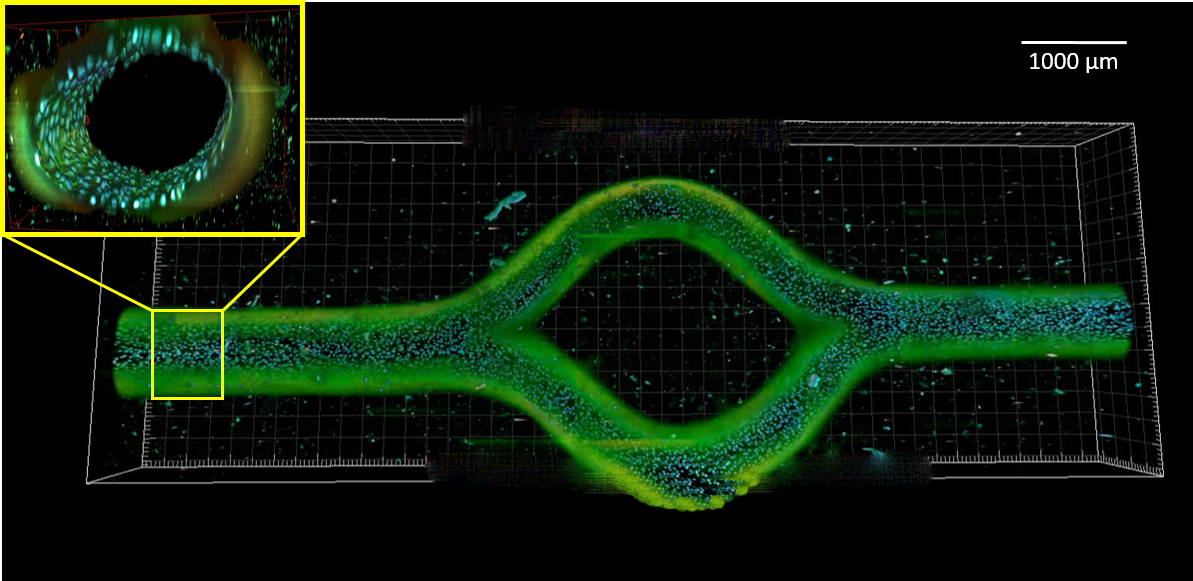
Current methods of microvessel fabrication mostly rely on self-assembly in tiny volumes, prefabricated designs of microchannels, or demand complicated multi-step procedures based on sacrificial templating to achieve specific geometrical features. Such application-oriented geometries subsequently require a design-specific and functional perfusion system that must be developed in parallel. In this study, we present a facile and flexible approach for the integration of biomimetically branched microvasculature within bulk hydrogels. For this, sacrificial scaffolds of thermoresponsive poly(2-cyclopropyl-2-oxazoline) (PcycloPrOx) are created using Melt Electrowriting (MEW) in an optimized and predictable way and subsequently placed into a customized bioreactor system, which is then filled with a hydrogel precursor solution. Aqueous environment above the lower critical solution temperature (LCST) of PcycloPrOx at 25°C swells the polymer without dissolving it, resulting in fusion of filaments that were deposited onto each other (print-and-fuse approach). Accordingly, an adequate printing pathway design results in generating physiological-like branchings and channel volumes which approximate Murray’s law in the geometrical ratio between parent
and daughter vessels. After gel formation, a temperature decrease below the LCST produces interconnected microchannels with distinct inlet and outlet regions. Initial placement of the sacrificial scaffolds in the bioreactors in a pre-defined manner directly yields perfusable structures via leakage-free fluid connections in a reproducible one-step procedure. Using this approach, rapid formation of a tight and biologically functional endothelial layer, as assessed not only through fluorescent dye diffusion, but also by tumour necrosis factor alpha (TNF-α) stimulation, is obtained within three days.






